13. A copper ball of mass m and specific heat c is at a temperature T1 = 86.85°C. The ball is thrown into a large lake at T2 = 6.85°C, which stays constant. What is the total change in entropy of the copper ball and the lake?
(A)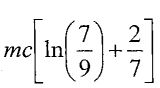
(B)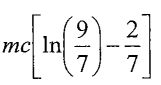
(C)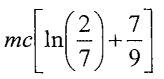
(D)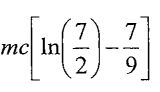
(E)None of the above
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料