20.同狀況條件下擴散4克CH4,需120秒,若擴散等重的SO2需多少秒?
(A) 480
(B) 240
(C) 120
(D) 60
答案:登入後查看
統計: A(10), B(23), C(12), D(32), E(0) #670153
統計: A(10), B(23), C(12), D(32), E(0) #670153
詳解 (共 2 筆)
#3139701
( 4 / 16 )0.5 = 0.5
( 4 / 64 )0.5 = 0.25
時間與分子量成開根號反比
120 * (0.25/0.5) = 60(s)
11
2
#6888526
MSO2=64.06g/mol
MCH4=16.05g/mol
Graham's law
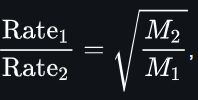
rate ∝ time-1
→ time2/time1=time2/120=(64.06/16.05)0.5=2
→ time2=240(s)
所以答案應選(B)
0
0