22. Consider the reaction 3A + B + C → D + E where the rate law is defined as
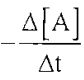
An experiment is carried out where [B]0 = [C]0 = 1.00 M and [A]0 = 1.00 × 10⁻⁴ M, and [A] = 3.26 × 10⁻⁵ M after 3.00 minutes. What is the half-life for this experiment?
(A)111 s
(B)87 s
(C)60 s
(D)17 s
(E)none of these
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料