30. Which of the following result(s) in an increase in the entropy of the system?
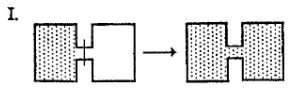
I. (See diagram.) II. \( \text{Br}_2(g) \rightarrow \text{Br}_2(l) \) III. \( \text{NaBr}(s) \rightarrow \text{Na}^+(aq) + \text{Br}^-(aq) \) IV. \( \text{O}_2(298\text{ K}) \rightarrow \text{O}_2(373\text{ K}) \) V. \( \text{NH}_3(1\text{ atm}, 298\text{ K}) \rightarrow \text{NH}_3(3\text{ atm}, 298\text{ K}) \)
(A)I
(B)II, V
(C)I, III, IV
(D)I, II, III, IV
(E)I, II, III, V
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料