34. The following data were obtained for the reaction of NO with \( O_2 \). Concentrations are in molecules/cm³ and rates are in molecules/cm³·s.
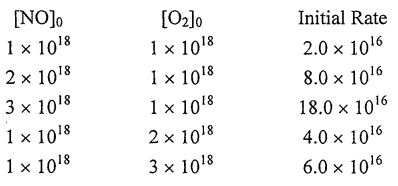
Which of the following is the correct rate law?
(A) Rate = \( k[NO][O_2] \)
(B) Rate = \( k[NO]^2 \)
(C) Rate = \( k[NO][O_2]^2 \)
(D) Rate = \( k[NO]^2[O_2] \)
(E) Rate = \( k[NO]^2[O_2]^2 \)
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料