7. Consider the states of a negatively charged hydrogen ion 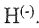 . Which of the following states violates the Pauli exclusion principle?
. Which of the following states violates the Pauli exclusion principle?
(A)2 electrons both occupying the 1s orbital and carrying opposite spins
(B) 1 electron occupying 1s and the other 2s and both carrying the same spin
(C) 1 electron occupying 1s and the other 2s and both carrying opposite spins
(D) 2 electrons both occupying 2px and carrying the same spin
(E) None of the above
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料