8. A Lewis dot structure for benzene, CH would give two equivalent resonance structures for this cyclic molecule with alternating single and double bonds
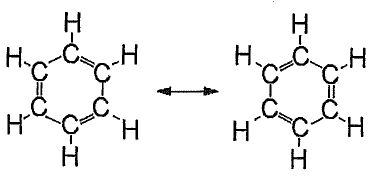
A combined LCAO and MO picture for the bonding of the carbon atoms in benzene would be
(A) 10(Csp²-Csp²) bonds between each C atom, and 3π MO delocalized over the C's
(B) 20(Csp3-Csp³) bonds between each C atom, and 1 7π MO delocalized over the C's
(C) 10(Csp³ Csp³) bonds between each C atom, and 1tMO delocalized over the C's
(D)1(Csp2-Csp²) bonds between each C atom, and 21 MO delocalized over theC's
(E) 30(C2p-C2p) bonds between each C atom
答案:登入後查看
統計: A(1), B(0), C(0), D(0), E(0) #3477613
統計: A(1), B(0), C(0), D(0), E(0) #3477613