12. For the synthesis of ammonia at 500°C, the equilibrium constant is 6.0 x 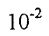 L²/mol². Predict the direction in which the system will shift to reach equilibrium in the following case.
L²/mol². Predict the direction in which the system will shift to reach equilibrium in the following case.
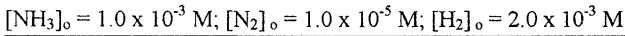
(A) The ammonia synthesis reaction will go backward.
(B) The ammonia synthesis reaction will go forward.
(C) The ammonia synthesis reaction is under equilibrium.
(D) The ammonia synthesis reaction is under equilibrium and will stop reaction.
(E) The ammonia synthesis reaction will not work.
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料