2. For a reaction at constant pressure, the standard Gibbs energies at two temperatures are: ΔrG°(298K) = −12.0 kJ mol⁻¹, ΔrG°(348K) = −5.0 kJ mol⁻¹. Using the Gibbs-Helmholtz equation: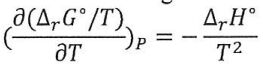
what can be concluded about the sign of ΔrH°? (assuming that ΔrH° does not depend on T)
(A) ΔrH° = 0
(B) ΔrH° > 0 (endothermic) and ΔrH° is around +54 kJ mol⁻¹
(C) ΔrH° < 0 (exothermic)
(D) Cannot be inferred from ΔrG°(T)
(E) ΔrH° > 0 (endothermic) and ΔrH° is around +5.4 kJ mol⁻¹
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料