4. A reaction proceeds by the mechanism: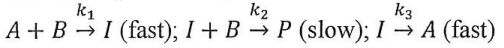 Assuming the steady-state approximation for the intermediate I (i.e., d[I]/dt ≈ 0), what is the rate law for product formation r =
Assuming the steady-state approximation for the intermediate I (i.e., d[I]/dt ≈ 0), what is the rate law for product formation r =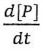 in terms of [A] and [B]?
in terms of [A] and [B]?
(A) r = k2[A][B]
(B) 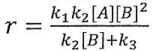
(C)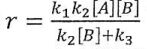
(D) r = k1[A][B]
(E) r = k2[I][B]
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料