23. The energy expressions for the electrons in the He* ion and the hydrogen atom are

Which of the following statements is(are) correct?
I. For the transitions n1 →n2, the frequency is larger for H than for 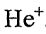 .
.
II. The first ionization energy of the H atom is smaller than the second ionization energy of the He atom.
III. The 1s orbital in 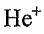 is larger (in the sense that the probability density is shifted outward) than the 1s orbital in H.
is larger (in the sense that the probability density is shifted outward) than the 1s orbital in H.
(A) I only
(B) II only
(C) III only
(D) I and II only
(E) I, II, and III
答案:登入後查看
統計: A(0), B(1), C(0), D(1), E(1) #3477628
統計: A(0), B(1), C(0), D(1), E(1) #3477628