3.碳酸鈣的標準莫耳分解熱為+1190 kJ,則在標準狀態時,下列何者正確?
(A) CaCO3(s) → CaO(s)+CO2(g),ΔH=-1190 kJ
(B) CaCO3(s) → CaO(s)+CO2(g),ΔH=+1190 kJ
(C) CaCO3(s) → Ca(s)+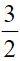 O2(g)+C(s)+1190 kJ
O2(g)+C(s)+1190 kJ
(D) CaCO3(s)+1190 kJ → Ca(s)+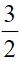 O2(g)+C(s)
O2(g)+C(s)
(E) CaCO3(s) → Ca2+(aq)+CO32-(aq),ΔH=+1190 kJ
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料