7. . Consider the formation of MgO( s) • Assume that △H° and △S° are independent of temperature. Calculate the temperature at which the formation of MgO switches. from spontaneous to non-spontaneous or vice versa?
[Hint: Mg(s) 十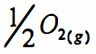 ➔MgO(s) ;△H° = -602kJ I mo!; △S° = -1081 I K · mol]
➔MgO(s) ;△H° = -602kJ I mo!; △S° = -1081 I K · mol]
(A)2,574K,
(B)3,574K,
(C)4,574K,
(D)5,574K,
(E) none of above
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料