9.The equilibrium constant for an association reaction A+B 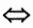 ABis 1.8x103 dm³ / mol at 25°C and 3.45×103 dm³ / molat 40°C. Assuming AHto be independent of temperature, calculate △H0=? [Hint:
ABis 1.8x103 dm³ / mol at 25°C and 3.45×103 dm³ / molat 40°C. Assuming AHto be independent of temperature, calculate △H0=? [Hint: 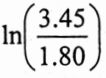 =0.651
=0.651
(A)33.67 J/mol;
(B) 336.7 J/mol;
(C) 3367 J/mol;
(D) 33.67 kJ/mol;
(E) 336.7 kJ/mol;
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料