70. Three types of chemical reactions are described below. Which of the following statements is correct?
I. CH₃COOH(l) + CH₃CH₂OH(l) 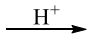 CH₃COOCH₂CH₃(l) + H₂O
CH₃COOCH₂CH₃(l) + H₂O
II. AgNO₃(aq) + KCl(aq) → AgCl(s) + K⁺(aq) + NO₃⁻(aq)
III. Cu(s) + 2Fe³⁺(aq) → Cu²⁺(aq) + 2Fe²⁺(aq)
(A) At 25 °C (1 atm), the reaction rate for type I is the fastest.
(B) This equation of type II is called the net ionic equation.
(C) Type III is a precipitation reaction.
(D) The reduced half-reaction of type III is Cu(s) → Cu²⁺ + 2e⁻.
(E) Type I involves bond cleavage and formation reaction.
答案:登入後查看
統計: A(0), B(0), C(0), D(1), E(0) #3853585
統計: A(0), B(0), C(0), D(1), E(0) #3853585