6. 1 mol氮氣理想氣體,由初始狀態 T1 = 300 K、 P1 = 1×105 Pa改變至T2 = 450 K、P2 = 2×105 Pa ,已知定壓莫耳熱容量Cp =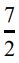 R並視為定值,請問氮氣的熵變化(ΔS)為多少?
R並視為定值,請問氮氣的熵變化(ΔS)為多少?
(A) 5.76 J/K
(B) 6.04 J/K
(C) 8.43 J/K
(D) 11.80 J/K
答案:登入後查看
統計: A(0), B(5), C(0), D(0), E(0) #3803844
統計: A(0), B(5), C(0), D(0), E(0) #3803844