6. Chlorobenzene and bromobenzene have similar structures and molecular dipole moments. Therefore, these two compounds can form an ideal solution. Given that the vapor pressures of pure chlorobenzene and bromobenzene at 25°C are 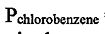 = 8.8 Torr,
= 8.8 Torr, 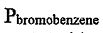 = 4.2 Torr, respectively, calculate the molar fraction of bromobenzene in the vapor phase for a 1:4 chlorobenzene/bromobenzene solution.
= 4.2 Torr, respectively, calculate the molar fraction of bromobenzene in the vapor phase for a 1:4 chlorobenzene/bromobenzene solution.
(A)0.20
(E) 0.86
(B) 0.34
(C) 0.66
(D) 0.80
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料