題組內容
1. A 17.5 g mass of ice at 273 K is added to 125 g of H2O (I) at 325 K at a
constant pressure. Assume 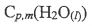 (heat capacity per mole at constant
pressure)=
(heat capacity per mole at constant
pressure)=  = 75.3 J/Kmol;
= 75.3 J/Kmol;  = 6010 J/mol.
= 6010 J/mol.
1. A 17.5 g mass of ice at 273 K is added to 125 g of H2O (I) at 325 K at a
constant pressure. Assume 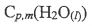 (heat capacity per mole at constant
pressure)=
(heat capacity per mole at constant
pressure)=  = 75.3 J/Kmol;
= 75.3 J/Kmol;  = 6010 J/mol.
= 6010 J/mol.