題組內容
1. Titanium dioxide is a wide-bandgap seniconductor that is considered as an insulating dielectric
in VLSI capacitors and for use in solar applications such as solar cells and photocatalyst. Thin
films of TiO2 can be prepared using chemical vapor deposition from gascous titanium
tetra-isopropoxide (TTIP). The overall reaction is
Ti(OC3H7)4 → TO2+4C3H6+2H2O
The reaction mechanism in a CVD is believed to be
TTIP (g) + TTIP (g) 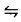 I(g) + P (g)
I(g) + P (g)
I(g)+S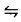 I.S
I.S
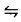 I(g) + P (g)
I(g) + P (g) I(g)+S
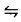 I.S
I.S I.S → TiO2+ P2(g)
Where I is an active intermediate, S is an active site, Pi is one set of reaction products (e.g. H2O,
C3H6) and P2 is another set. Assuming the homogeneous gas-phase reaction for TTIP is in
equilibrium, please answer the following question
(b) (8%) The experimental results show that at 200℃ the reaction is second order at low partial pressure of TTIP and zero order at high partial pressures. While at higher temperature (300 ℃ ) the reaction is second order in TTIP over the entire pressure range. Discuss these results based on the rate law derived above. [Hint: adsorption is exothermic in general]