10. Allyl radical (CH2-CH-CH2) is a planar molecular radical in which the three a electrons are delocalized. Hückel theory is a simplified molecular orbital (MO) theory describing the MOs of the n-electron system as linear combinations of the 2pz atomic orbitals at each carbon atom involved in the delocalized system. The basic assumptions of Hückel theory are: All overlap integrals are set equal to zero; All resonance integrals between non-neighboring atoms are set equal to zero; All coulomb integrals are set equal to a; All resonance integrals between neighboring atoms are set equal to β. What is the total energy of the three πelectrons predicted by Hückel MO theory?
(A)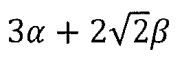
(B)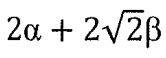
(C)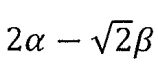
(D) 2a +4β
(E) 4α +4β
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料