8. The hydrogen atomic wave functions can be expressed as 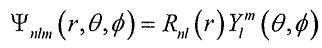 , where
, where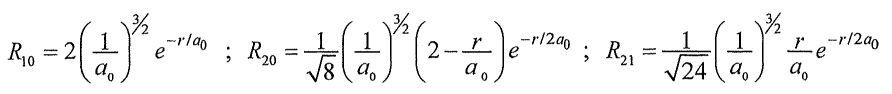
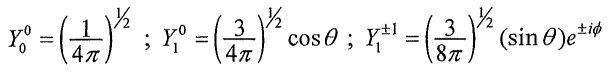 An electron is in the 2p0 orbital of a H atom. What is the most probable radius (the radius where the radial distribution function is at maximum) of finding that electron?
An electron is in the 2p0 orbital of a H atom. What is the most probable radius (the radius where the radial distribution function is at maximum) of finding that electron?
(A) 5a0
(B) 4a0
(C) 3a0
(D) 2a0(Ε) a0
答案:登入後查看
統計: 尚無統計資料
統計: 尚無統計資料