2. (a) Show that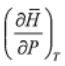 = 0 for an ideal gas, where
= 0 for an ideal gas, where 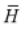 and P are molar enthalpy and pressure,
and P are molar enthalpy and pressure,
respectively. You can start from the equation below 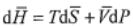
where 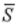 is molar entropy,
is molar entropy, 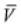 is molar volume, and T is temperature
is molar volume, and T is temperature
2. (a) Show that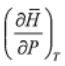 = 0 for an ideal gas, where
= 0 for an ideal gas, where 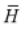 and P are molar enthalpy and pressure,
and P are molar enthalpy and pressure,
respectively. You can start from the equation below 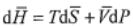
where 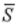 is molar entropy,
is molar entropy, 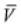 is molar volume, and T is temperature
is molar volume, and T is temperature